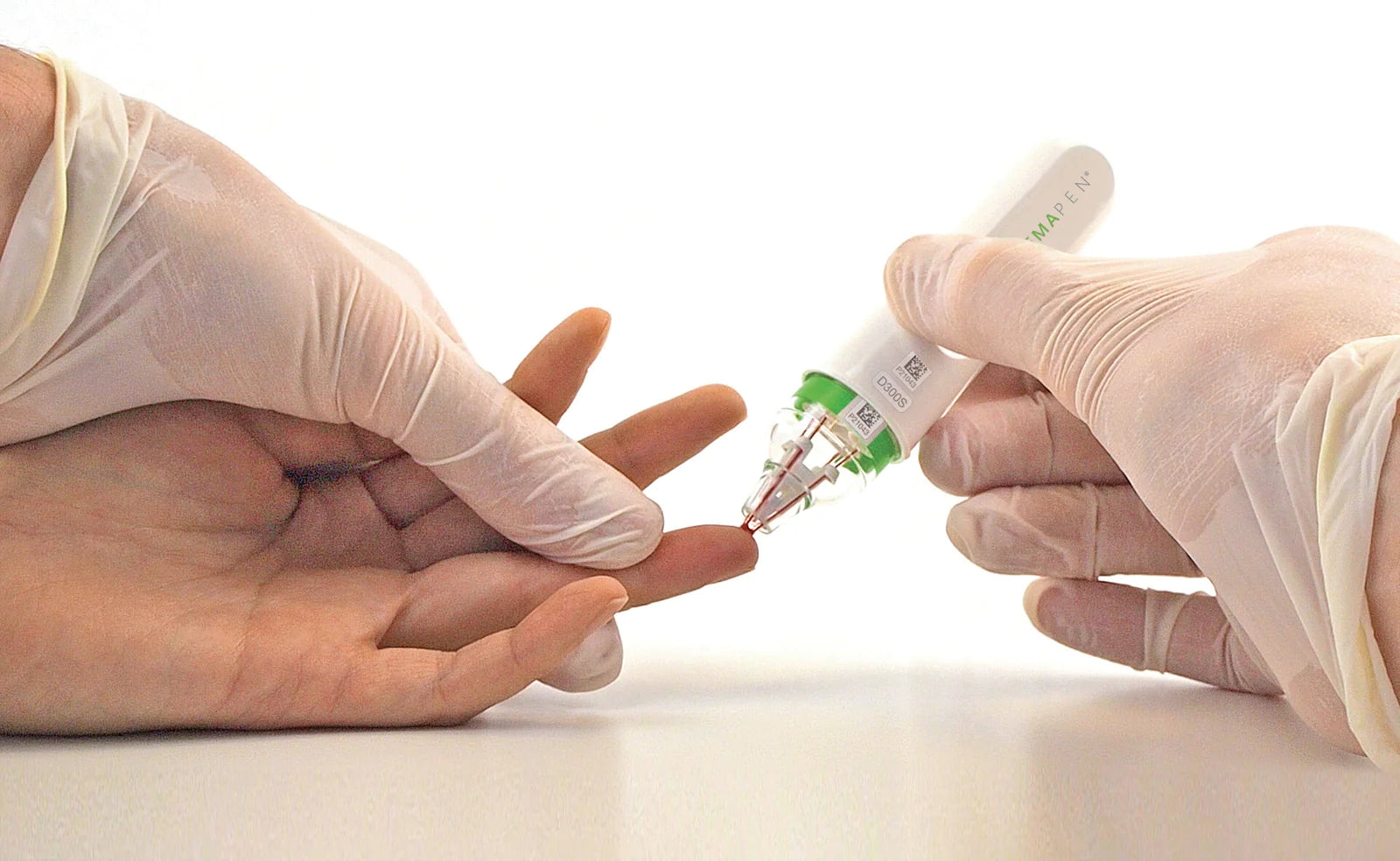
Trajan Scientific and Medical has listed its hemaPEN® blood microsampling device with the U.S. Food and Drug Administration as Class I for therapeutic and in vitro diagnostic use. This follows the ...
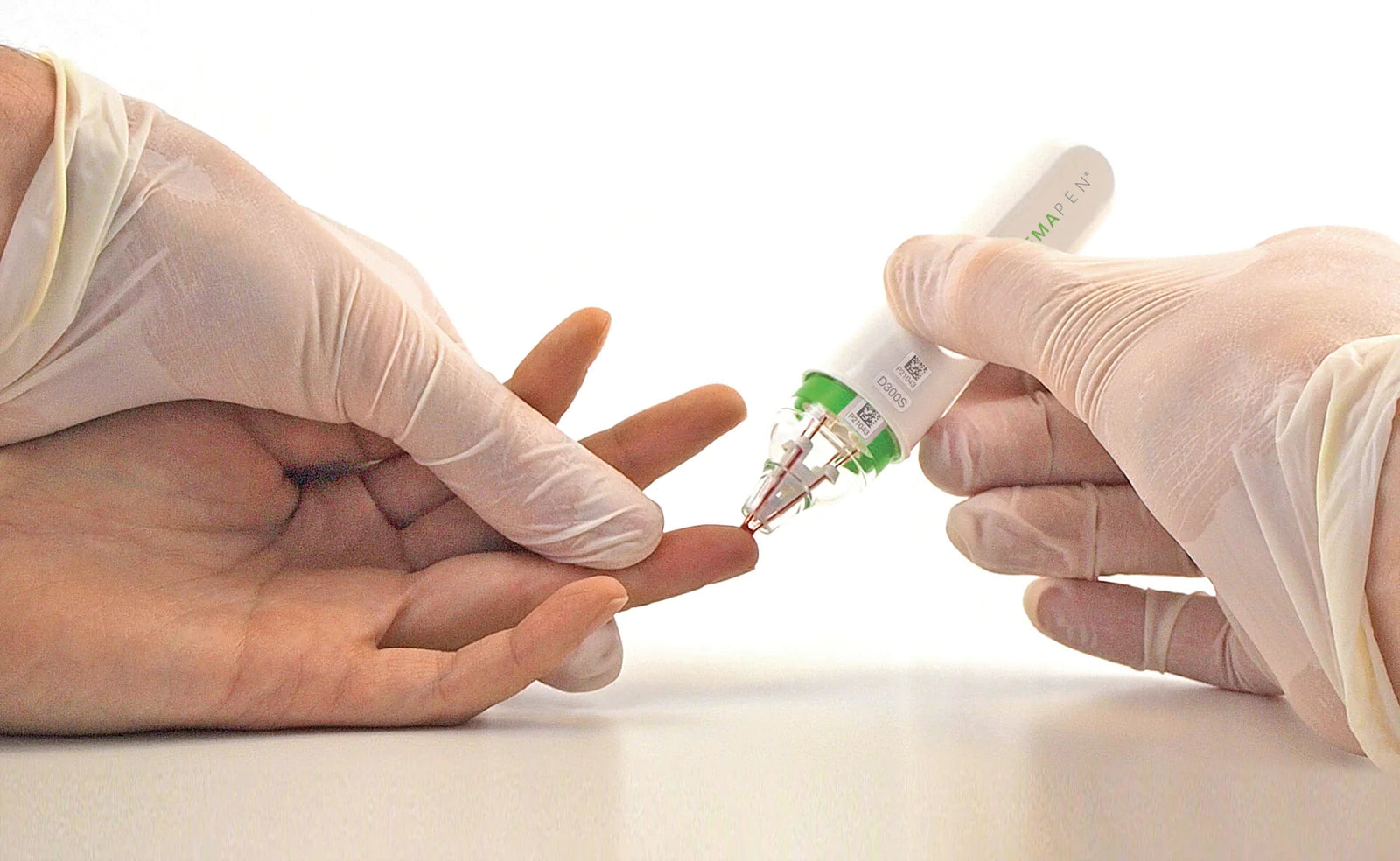
Trajan Scientific and Medical has declared hemaPEN compliance with EU IVD Directive 98/79/EC, and its release for in vitro diagnostic use in the EU and UK. This follows the hemaPEN’s recent additio...

Trajan Scientific and Medical releases new microtome blades in Australia, New Zealand, UK and EU, to provide the blade of choice for any tissue type. For histologists who have trouble finding a bla...

Blood microsampling technology has been evolving over the past few years. Designed to cut the need for central pathology collection centers and trained phlebotomists. It enables the collection of v...

Trajan Scientific and Medical and Queen’s University, Kingston, Canada, have been awarded a Mitacs Accelerate grant to develop new multiple electrospray (MES) technologies to increase throughput an...

Blood testing technology developed by South Australian medical researchers is poised to transform the nutrition industry. Trajan Nutrition CEO Marco Baccanti said, “Pathology labs can also now star...

Trajan Nutrition has signed a Memorandum of Understanding with the South Australian Health and Medical Research Institute (SAHMRI) to accelerate the translation in the global market of nutrition te...