Trajan Scientific and Medical (Trajan) is proud to advise that the European Patent Office will be granting a patent for hemaPEN® as a liquid collection device in Belgium, France, Germany, Ireland, Luxembourg, Monaco, Switzerland, Liechtenstein and the United Kingdom. Extensions are being sought across the remainder of Europe.
This follows the recent listings of hemaPEN with the U.S. Food and Drug Administration (US FDA) as Class I for therapeutic and in vitro diagnostic (IVD) use, with the Australian Register of Therapeutic Goods (TGA ARTG) as a Class 1 IVD, and it’s compliance with EU IVD Directive 98/79/EC for supply in the EU and UK as a General IVD.
In today’s environment, where vulnerable communities are practising self-isolation due to COVID-19, remote microsampling facilitates ongoing blood monitoring of chronic disease, and enables continuity-of-care.
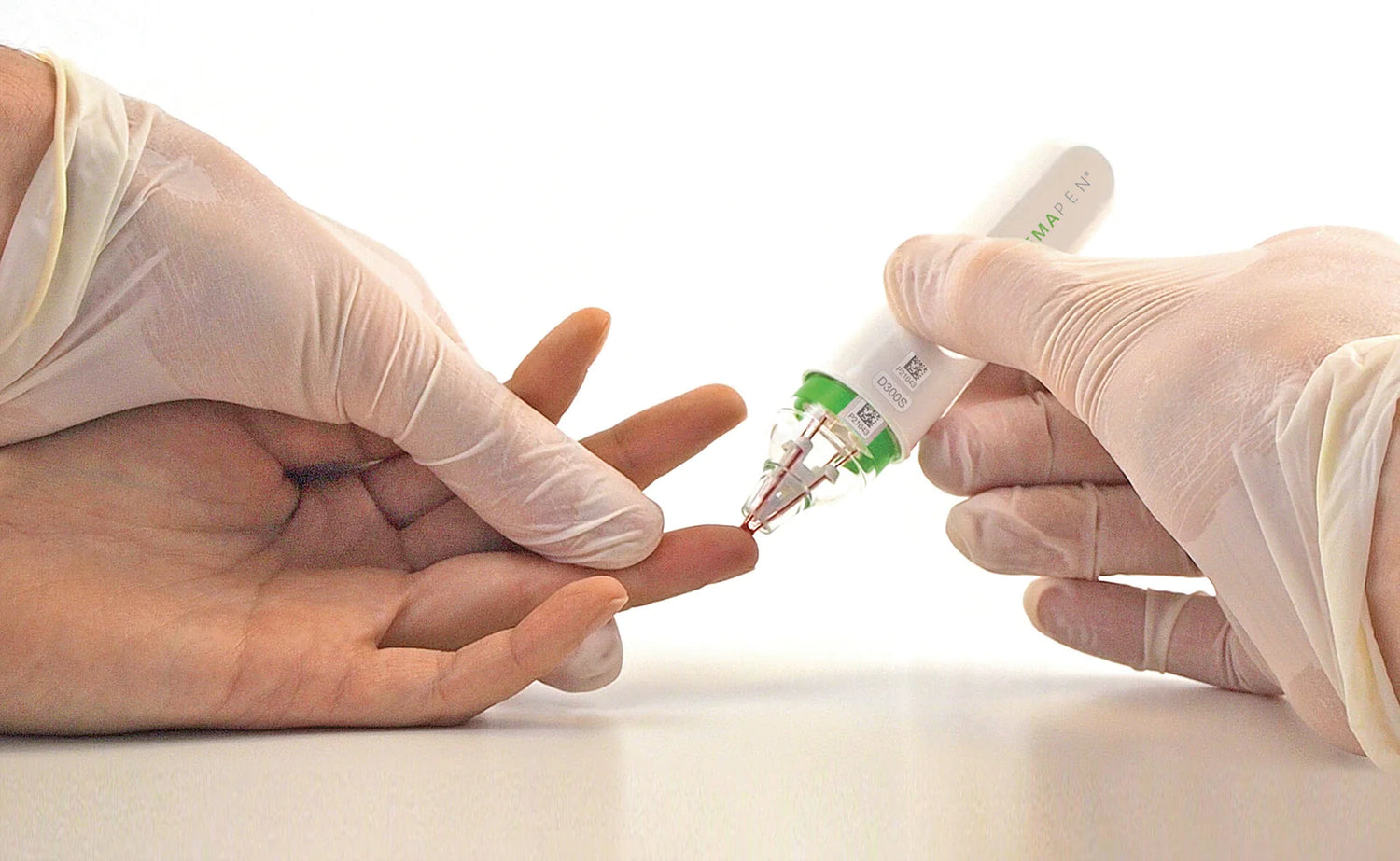
hemaPEN provides a convenient sampling procedure for collection and storage of four dried blood spot (DBS) samples. Unlike conventional DBS sampling tools, hemaPEN enables collection of an accurate and precise fixed micro-volume and is designed to maintain sample integrity for quantitative analysis. It is an easy-to-use sophisticated microsampling tool in the hands of non-analysts.
Trajan’s CEO, Mr Stephen Tomisich said, “Having a patent granted for hemaPEN will highlight the unique innovation and technology that is the hemaPEN device. We continue to be motivated by the interest shown in hemaPEN, and how it is being used to transform blood collection and storage.”
Trajan believes in science that benefits people - creating portable and affordable measurement solutions, enabling accurate results to inform preventative healthcare.
Visit www.hemapen.com to purchase or learn more about hemaPEN, and to sign up for updates.
To learn more about Trajan’s microsampling technologies and capabilities visit www.trajanscimed.com/microsampling.
 Download
Download
Press release [PDF]
Related news
The third leg in at-home healthcare – microsampling
US FDA listing of Trajan’s hemaPEN® blood microsampling device
CE mark for Trajan’s hemaPEN blood microsampling device, now available for diagnostic use across EU and UK
Trajan’s hemaPEN included on TGA’s ARTG as first blood microsampling device for use in Australia
Pens and prospering as a manufacturer
More information
www.hemapen.com
Media contact information
Trajan Scientific and Medical
media@trajanscimed.com